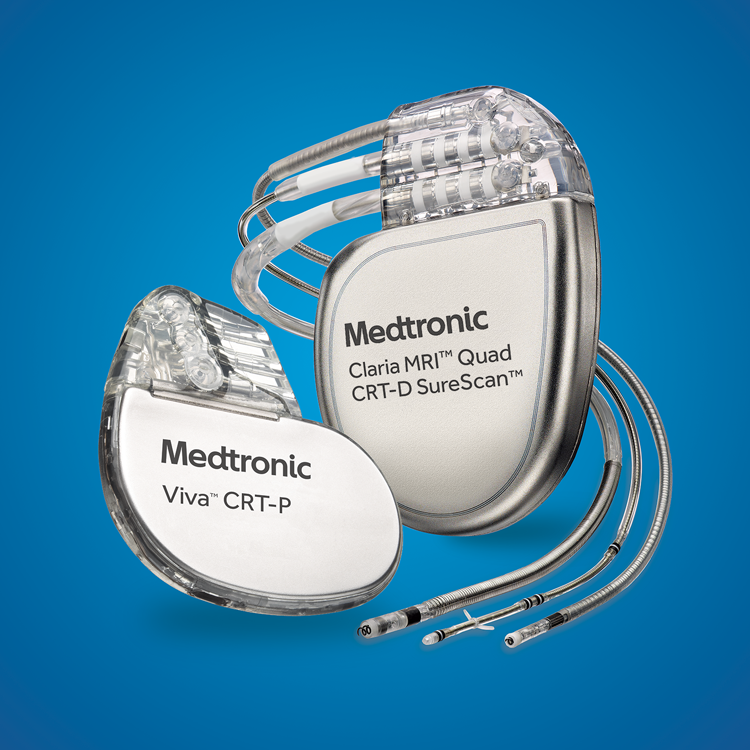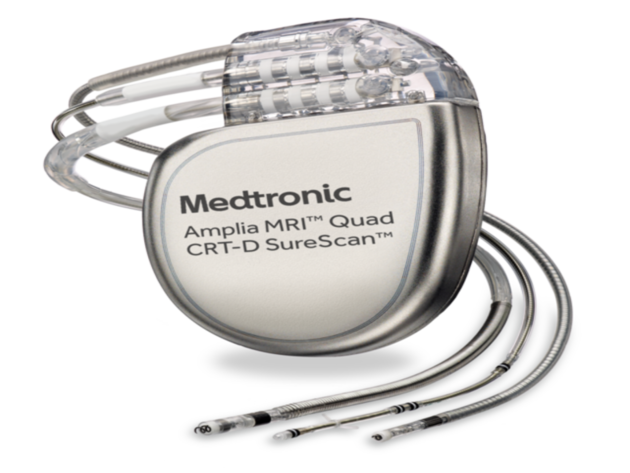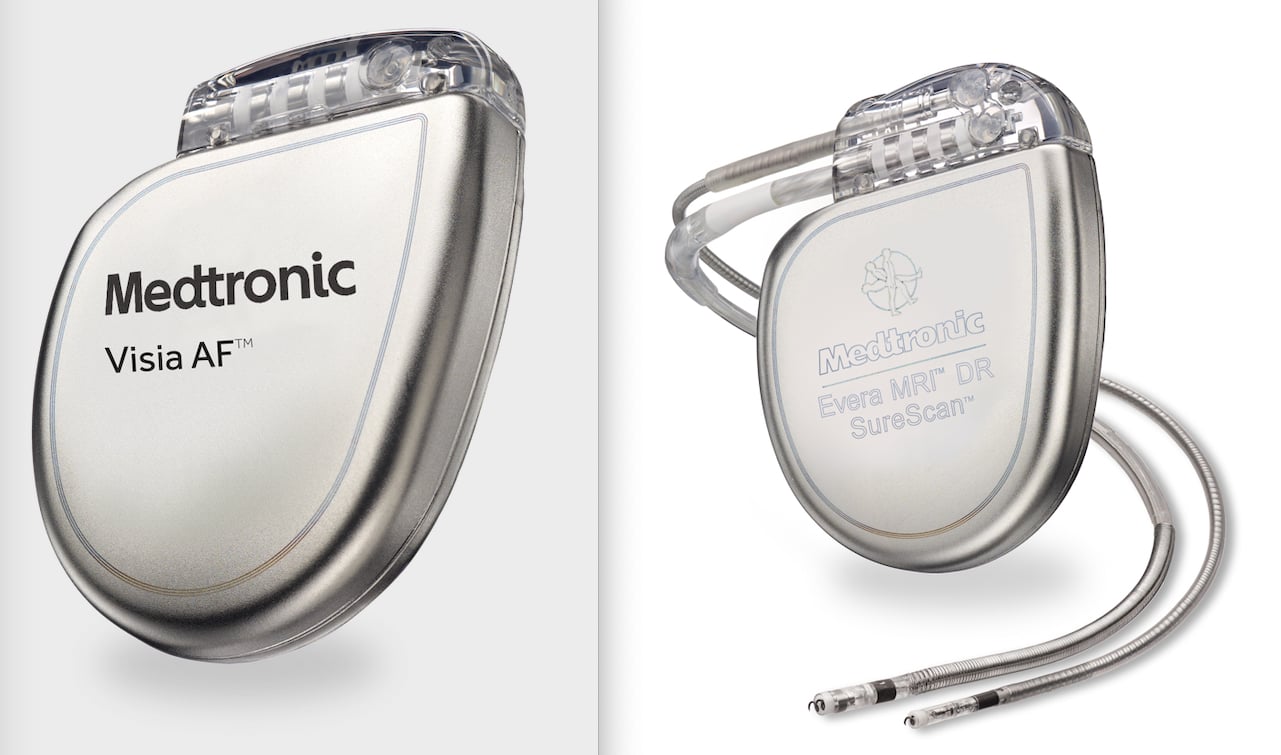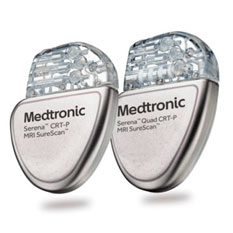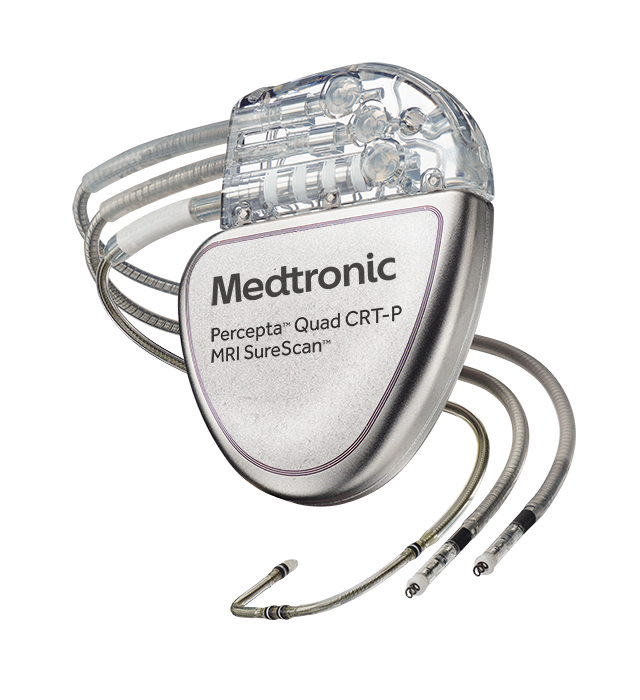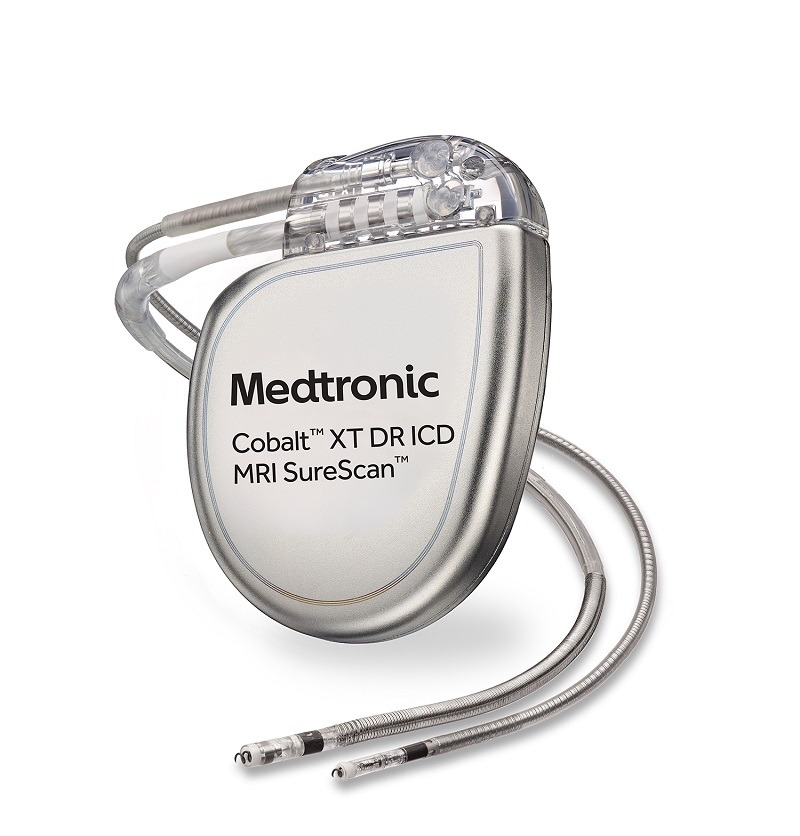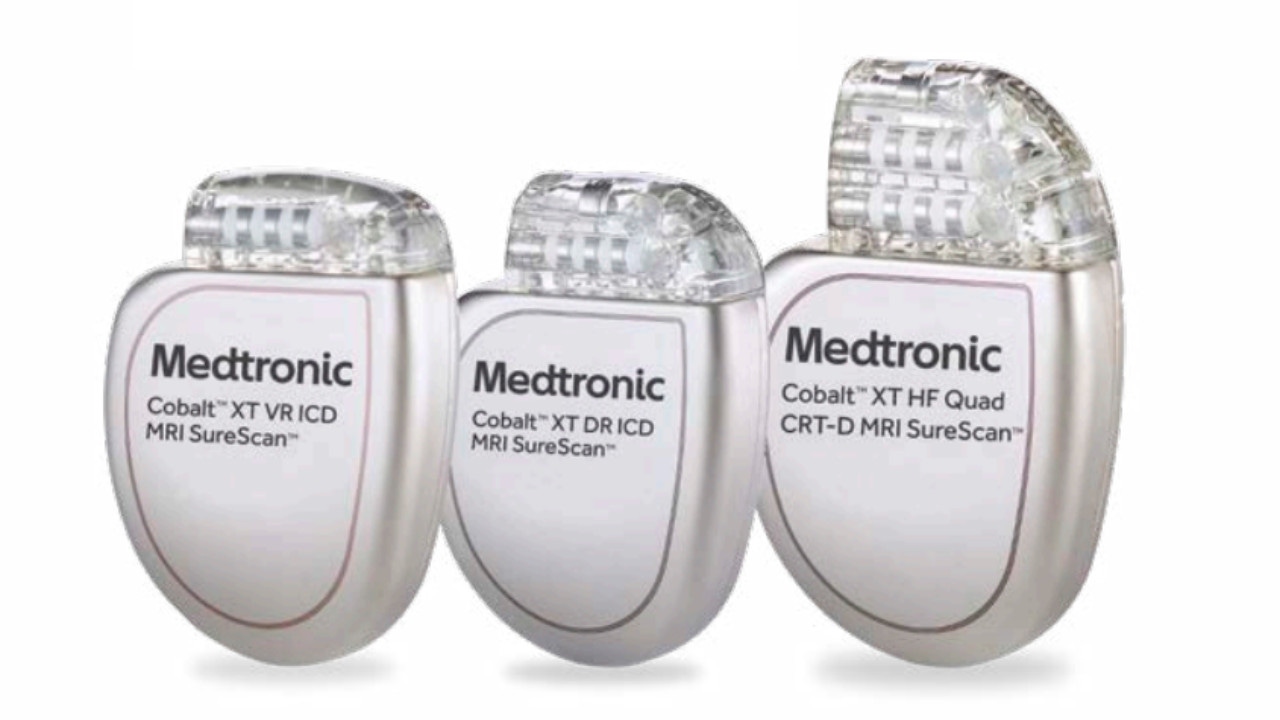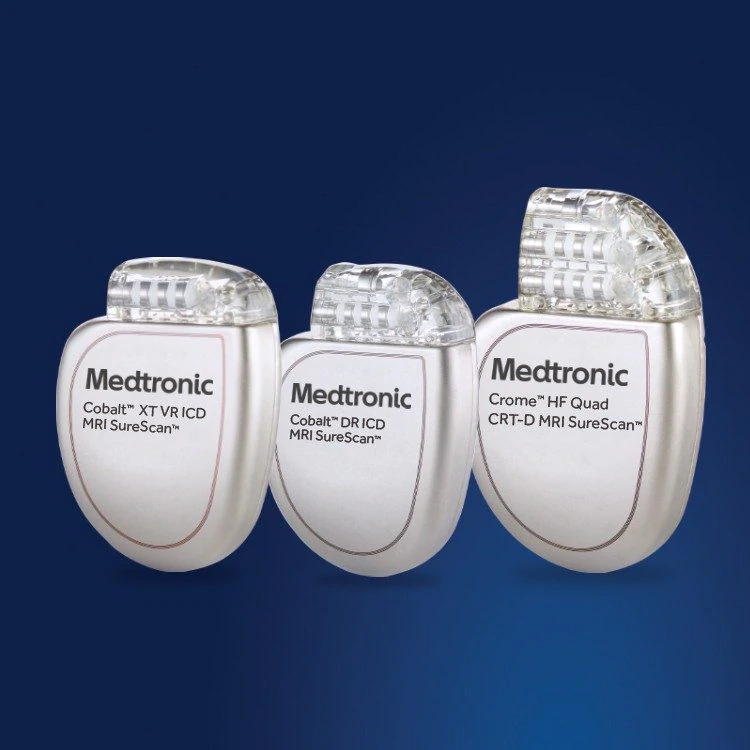
FDA announces recall of nearly 88,000 implantable cardiac devices due to risk of serious injury or death

Medtronic India - The Cardiac Resynchronisation Therapy is a clinically proven treatment for certain patients with heart failure. It improves the heart's ability to pump blood and oxygen to the body with